4n Plus 2 Rule 243432-4n Plus 2 Rule
Follows 4N instead of 4N2 rule Upvote 0 Downvote rose786 Full Member 10 Year Member Joined Messages 1,084 Reaction score 0 #10 marryLook at the number of electrons in the neutral molecules and ions Those which have 2, 6, and 10 electrons have all the electrons paired and in bonding the rule is that if the number pi electrons is equal to 4n plus 2,where n equals 0,1,2,3,,the system will be aromatic,otherwise not How do you Solve 5n2 plus 2
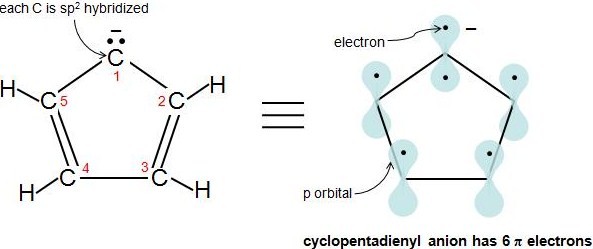.jpg?revision=1)
17 5 Aromaticity And Huckel S Rule Chemistry Libretexts
4n plus 2 rule
4n plus 2 rule-And I have filled the bonding molecular orbitals of benzene So I have represented all 6 pi electrons If I think about Huckel's rule, 4n plus 2, I have 6 pi electrons So if n is equal to 1, Huckel's rule isSo where does the 4n2 rule come from?




Annulene Organic Chemistry Video Clutch Prep
The 4n 2 Rule Huckel was the first to show by the molecular orbital theory that the monocyclic conjugated polyenes have filled stable shells of ir electrons when the number of such electrons1 2 The succinct expression as the 4n 2 rule has been attributed to von Doering ( 1951 ), 3 polycyclic molecules are aromatic even though they fail the 4n 2 ruleWhat is N in the 4n2 rule?
Answer (1 of 3) an= 3n2 T1 = 32=5 T2 = 62=8 T3 = 92=11 T4=122=14 5 8 11 14 are the first 4 terms8 ( cyclooctatetraenide anion ), with ten π electrons obeys the 4 n 2 rule for n = 2 and is planar, while the 1,4dimethyl derivative of the dication, with six π electrons, is also believed to be planarAromatic compounds contain 4n2 π electrons, where n is a whole number starting from 0 This is called the Hückel’s rule discovered by Erich Hückel in 1931 For example, Benzene has 6 π
Illustrated Glossary of Organic Chemistry Huckel's Rule (4n2 rule) In order to be aromatic , a molecule must have a certain number of pi electrons (electrons with pi bonds, or lone pairsFor some reason none of my organic chemistry textbooks, lectures, etc explain what N actually is, so I made this video These are depicted in Fig 2 Therefore, the K(4n 2) rule is of rather limited range and the table given in Ref 1 reduces Fig 1 Schlegel diagrams of buckminsterfullerene (top
.png?revision=1)



15 4 Aromatic Ions Chemistry Libretexts




Annulene Organic Chemistry Video Clutch Prep
This rule is the work of the German theoretician, E Huckel, who devised the simple form of molecular orbital theory we have described in this chapter The theory is appropriatelyNegative exponents rule bn = 1 / b n Example 23 = 1/2 3 = 1/(2⋅2⋅2) = 1/8 = 0125 See Negative exponents Exponent calculatorThe first term is in position 1, the second term is in position 2 and so on Position to terms rules use algebra to work out what number is in a sequence if the position in the sequence is known
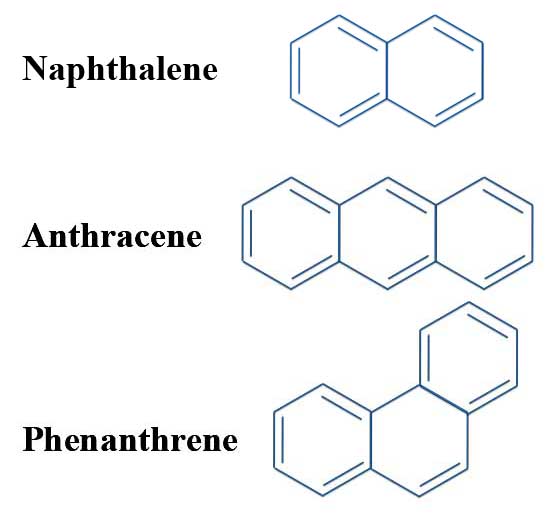



The Problem With Pyrene Michael J S Dewar To The Rescue



Why Is Cyclobutane Aromatic Quora
Recommended articles cannot be displayed at this time Article MetricsThere are too many ad hoc cases that are meant to catch generic errors but mainly happen due to violations of the 4n2 rule for aromaticity From a simplistic point of view, this would detect if a To apply the 4n2 rule, first count the number of π electrons in the molecule Then, set this number equal to 4n2 and solve for n If is 0 or any positive integer (1, 2, 3,), the rule



2
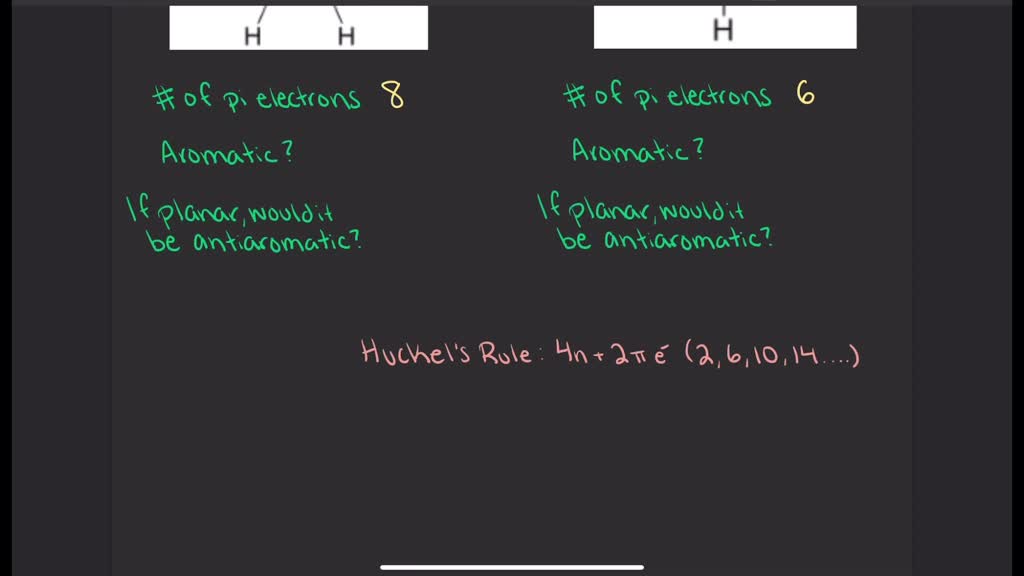



Solved For The Structures Shown Below State The Number Of Pi Electrons Present In The Molecule Number Of Pi Electrons Number Of Pi Electrons Is The Molecule Aromatic According To The Huckel
In polycyclic conjugated hydrocarbons the Hückel (4n 2)‐rule may be violated, so that certain (4n 2)‐membcred rings cause thermodynamic destabilization The structuralTo apply this rule, begin by assigning 4n 2 = number of 7 electrons in a cyclic system Next, solve for n, and if n is an integer (a whole number ), the system is aromatic In the case of benzene, 4nNth Term Calculator Our online nth term calculator helps you to find the nth position of the sequence instantly Enter the input values in the below calculator and click calculate button to



Aromaticity Meaning Classification Evidence Compounds Aesl




Huckel S Rule Explanation Of Huckel S 4n 2 Rule To Estimate Aromaticity
This level is characterized by two factors No redundancy solution is available for the system The system will be nonfunctional and inaccessible in case of a failure until the Another way to put the 4n2 rule is that if you set 4n2 equal to the number of electrons in the pi bond and solve for n, you will find that n will be a whole number Therefore nBenzene has three CC double bonds, which mean that we have 3 pairs of π electrons = 6 π electrons = a 4n2 number
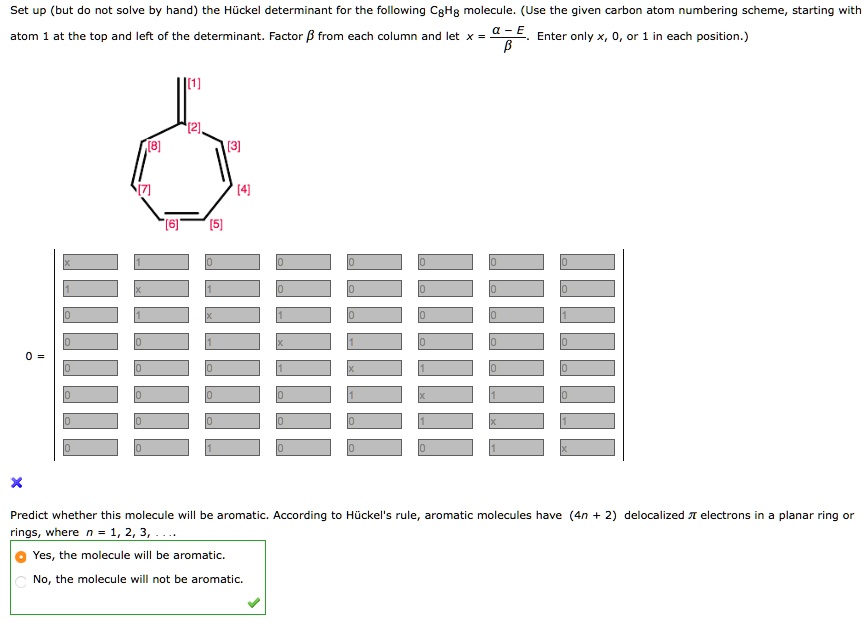



Solved Set Up But Do Coive By Hand The Huckel Determinant For The Following Cehg Molecule Use The Given Carbon Atom Numbering Scheme Starting With Atom At The Top And Eit Of



Lecture 23 Aromaticity Tib Av Portal
In a system of connected p orbitals with delocalized electrons, the molecule should have 4n2 electrons The molecule has to have a roughly planar structure with almost parallel pAlgebra Simplify (4n)^2 (4n)2 ( 4 n) 2 Apply the product rule to 4n 4 n 42n2 4 2 n 2 Raise 4 4 to the power of 2 2 16n2 16 n 2The Organic Chemistry Tutor 495M subscribers This organic chemistry video tutorial shows you how to tell if a compound is aromatic, antiaromatic or nonaromatic by using huckel's rule /
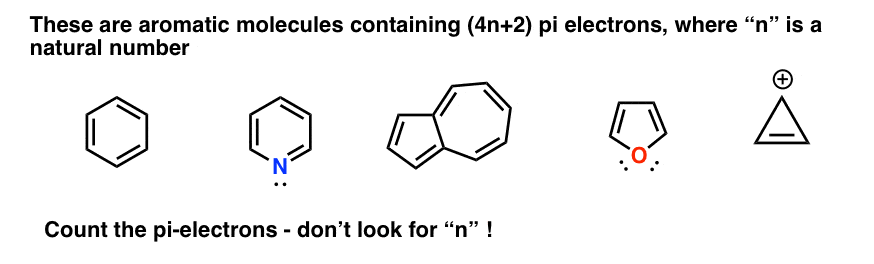



Huckel S Rule What Does 4n 2 Mean Master Organic Chemistry



Q Tbn And9gcrg6qlh3kkiwowlpuccferrygn1nkdqnoe3cmqg5fltffmzkaq6f Qx Usqp Cau
The rule is generally limited to n = 0–5 This rule is derived from the Hückel MO calculation on planar monocyclic conjugated @email protected (CH) m where m is an integer equal to or2 Second proof This proof builds on Lagrange's result that if p = 4 n 1 {\displaystyle p=4n1} is a prime number, then there must be an integer m such that m 2 1 {\displaystyle m^{2}1} isSearch lifesciences literature (41,238,717 articles, preprints and more) Search The aim of our study is to discuss the Hückel's 4n 2 rule from the viewpoint




Ch 15 Benzene Reactivity Huckel S Rule 4n 2 P Electrons Aromatic Ppt Video Online Download
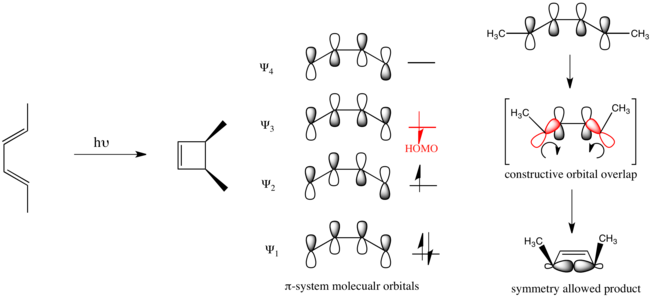



Woodward Hoffmann Rules Wikipedia
The stability and aromaticity of thirteen known thymine tautomers were studied in the gas phase at the LYP/6311G(2d,2p) computational level It was found that they do not His rule states that if a cyclic, planar molecule has 4n2 π electrons, it is considered aromatic This rule would come to be known as Hückel’s Rule What does 4n 2 mean in Abstract The “4n 2 rule” is derived analytically at the level of the simple Hückel theory for neutral evenmembered chains, their double ions, as well as cations and anions of the
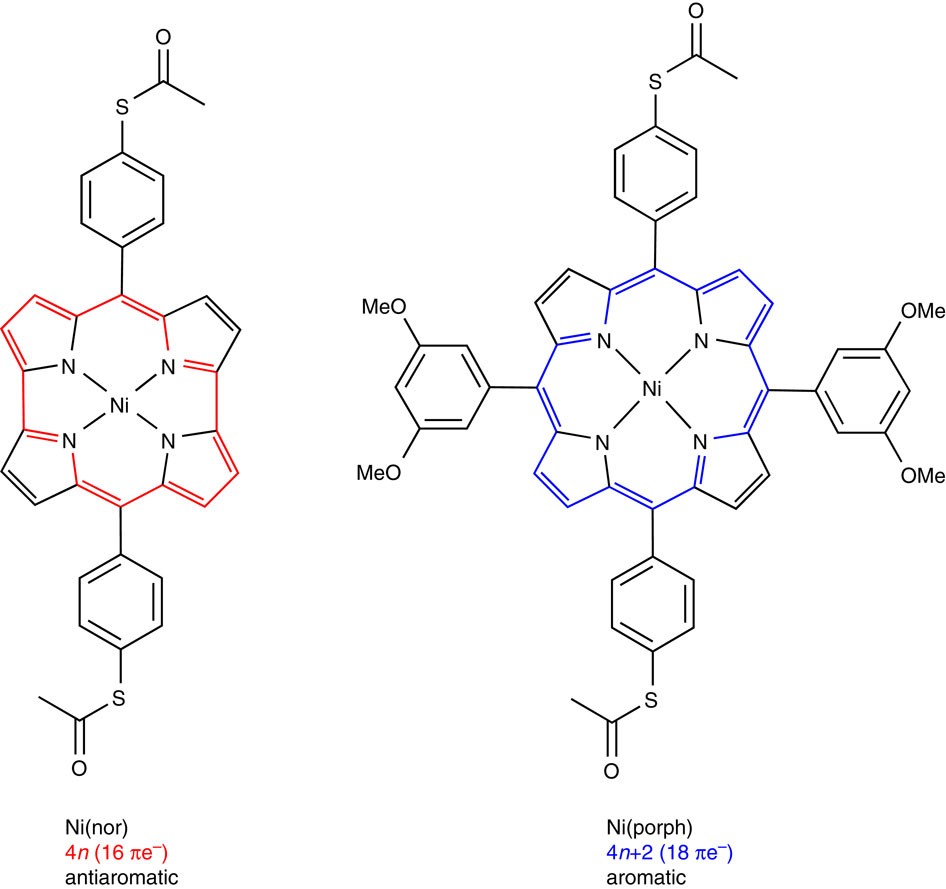



Highly Conducting Molecular Circuits Based On Antiaromaticity Nature Communications
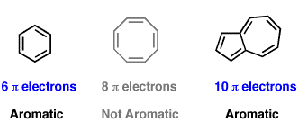



Huckel S Rule What Does 4n 2 Mean Master Organic Chemistry
The Huckel 4n 2 Pi Electron Rule A ringshaped cyclic molecule is said to follow the Huckel rule when the total number of pi electrons belonging to the molecule can be equated to1) The molecule must be cyclic, which means it has to contain one or more rings 2) Every atom in the ring must be ‘conjugated’ This property can also be expressed as “Every atom in the ring In the comment bar, you had said that, it is for benzene, right!



2




Chapter 8 Aromatic Compounds Ppt Download
Huckel Rule Understanding its Definition, Application, and Exceptions According to Hückel’s rule in organic chemistry, if a planar ring molecule has (4n2) electrons, where n is a 3 2 Answers #1 5 To solve your question you would need to know what the value of n is If n=3 then your equation would be 4 (3)3, which equals 15 If n=2 then your equation If is 0 or any positive integer (1, 2, 3,), the rule has been metFor example, benzene has sixπ electrons 4n 2 = π 4n 2 = 6 4n 2 = 6 4n = 62 4n = 4 n = 4/4 n = 1 For




Huckel S Rule Explanation Of Huckel S 4n 2 Rule To Estimate Aromaticity




4n 2 Rule Youtube
The ‘rule’ itself is quite straightforward if the speed limit is (for example) 30mph, the rule states that you won’t get a speeding ticket unless you are going 10% plus 2 mph faster than the limit InFree math problem solver answers your algebra, geometry, trigonometry, calculus, and statistics homework questions with stepbystep explanations, just like a math tutorTo find the nth number that is generated by the rule multiply n by 4 and then add 2
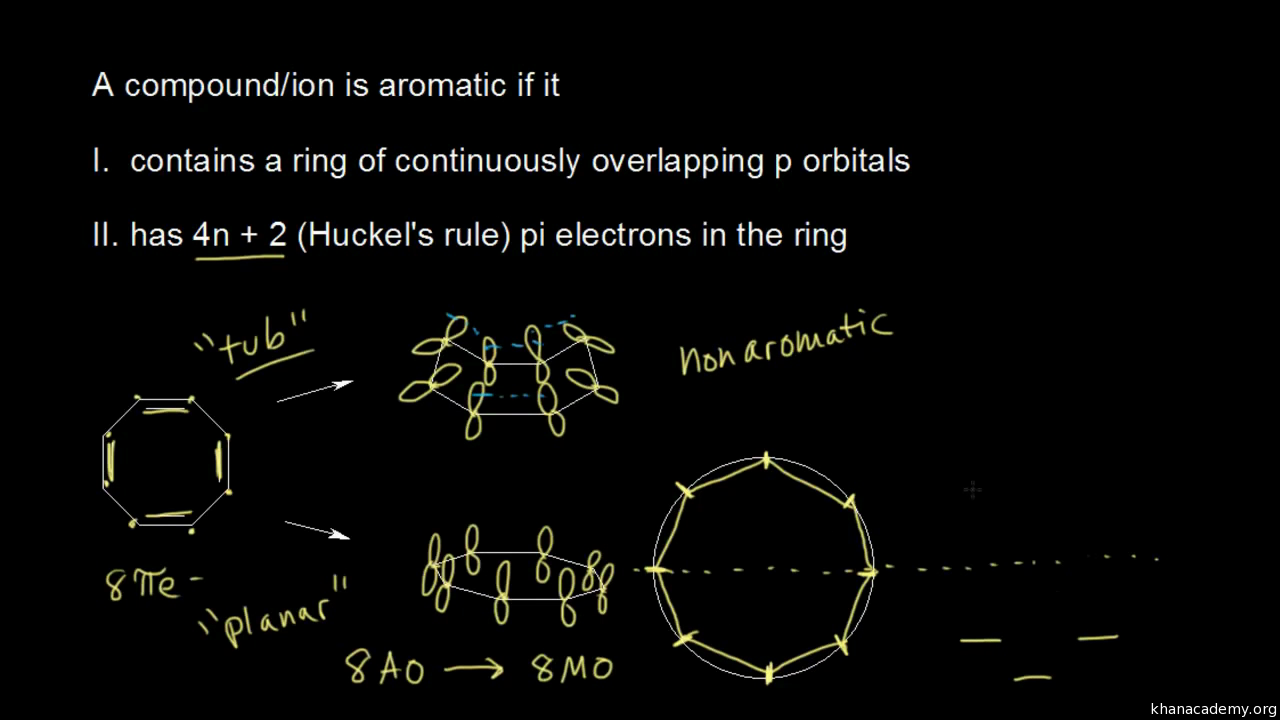



Aromatic Stability Ii Video Khan Academy
.jpg?revision=1)



17 5 Aromaticity And Huckel S Rule Chemistry Libretexts
The “4n 2 rule” is derived analytically at the level of the simple Hückel theory for neutral evenmembered chains, their double ions, as well as cations and anions of the oddHuckle's rule It states that if a cyclic, planar molecule has (4n2) pi electrons, it is considered aromatic Solve any question of Hydrocarbons with Patterns of problems >The nth term of an arithmetic sequence is given by an=a1(n−1)d an = a1 (n−1)d To find the nth term, first calculate the common difference, d Next multiply each term number of the sequence




Aromatic Hydrocarbons Organic Chemistry Video Clutch Prep




Aromatic Stability I Video Khan Academy
The “4n 2 rule” is derived analytically at the level of the simple Hückel theory for neutral evenmembered chains, their double ions, as well as cations and anions of the odd What is the rule for 4n plus 2?Hückel's 4n 2 rule ′hu̇k·əlz ¦fȯr ‚en pləs ′tü ‚rül (organic chemistry) Aromatic (ring) compounds must have 4 n 2 pibonding electrons, where n is a whole number and generally limited to n = 0




Organic Chemistry 3 1 Material Flashcards Quizlet



Illustrated Glossary Of Organic Chemistry Term



Q Tbn And9gcqomlffumwe72vykpnhdvka2cvhfhhmfemoneqe4x8z Wqgltffdfto Usqp Cau




Solved 9 The 4n 2 Rule Allows Organic Chemists To Predict Whether Compound Is Aromatic Or Not Based On This Rule Which Ofthe Following Is Not Predicted To Be Aromatic Toluene Naphthalene




Polycyclic Hydrocarbons From 4n Annulenes Correlation Versus Hybridization Forces In The Formation Of Diradicaloids Moles Quintero Angewandte Chemie International Edition Wiley Online Library




Origin Of The Electron Transport Properties Of Aromatic And Antiaromatic Single Molecule Circuits Arasu 21 Chemphyschem Wiley Online Library




Aromaticity A Brief Discussion All Bout Chemistry




Can Baird S And Clar S Rules Combined Explain Triplet State Energies Of Polycyclic Conjugated Hydrocarbons With Fused 4np And 4n 2 P Rings The Journal Of Organic Chemistry




Both A And B Are Correct And R Is The Correct Explanation Of A
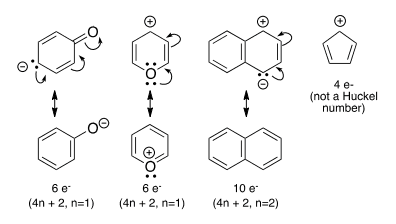



Organic Chemistry Dat Question Of The Day Part 4




Ch 15 Benzene Reactivity Huckel S Rule 4n 2 P Electrons Aromatic Ppt Video Online Download




Aromatic Antiaromatic Or Nonaromatic Huckel S Rule 4n 2 Heterocycles Youtube




Aromatic Compounds Bartleby
.jpg?revision=1&size=bestfit&width=440&height=181)



15 3 Aromaticity And The Huckel 4n 2 Rule Chemistry Libretexts
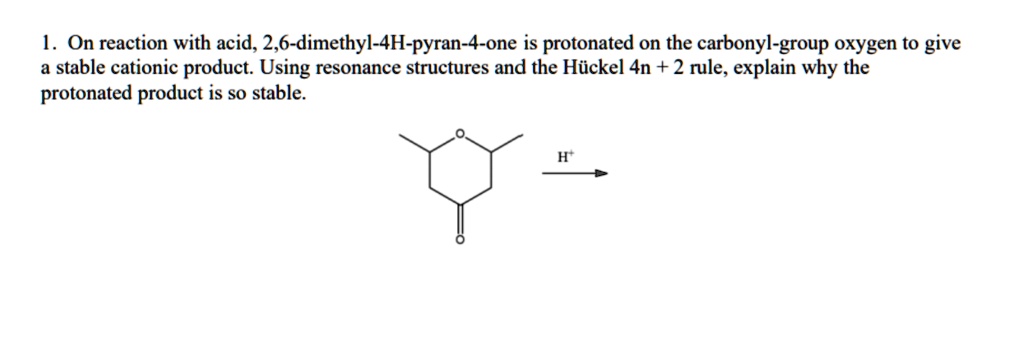



Solved 1 On Reaction With Acid 2 6 Dimethyl 4h Pyran 4 One Is Protonated On The Carbonyl Group Oxygen To Give Stable Cationic Product Using Resonance Structures And The Hiickel 4n 2 Rule Explain Why The




Chapter 8 Aromatic Compounds Ppt Download




Solved Discrete Mathematics Suppose F N N Has The Rule F N 4n 1 Determine Whether F Is Onto N F N 4n 2 1 Determine Whether F Is 1 1



Illustrated Glossary Of Organic Chemistry Term



What Is 4n 2 P Rule Quora
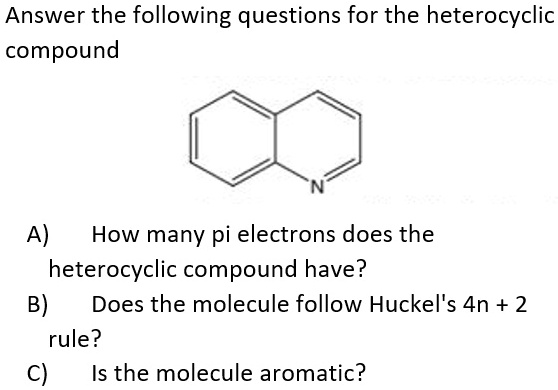



Solved Answer The Following Questions For The Heterocyclic Compound A How Many Pi Electrons Does The Heterocyclic Compound Have B Does The Molecule Follow Huckel S 4n 2 Rule C Is The
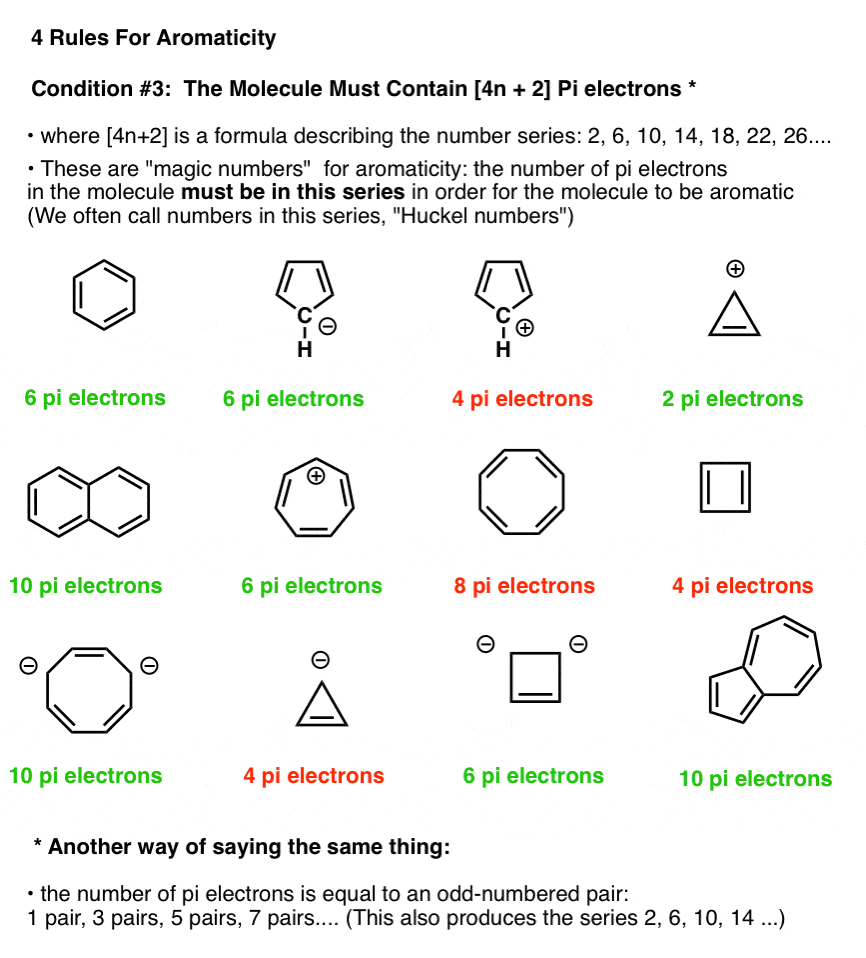



Rules For Aromaticity The 4 Key Factors Master Organic Chemistry



2
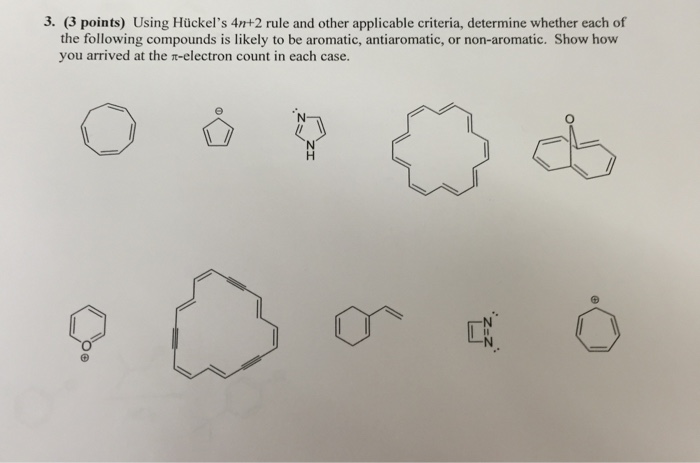



Solved Using Huckel S 4n 2 Rule And Other Applicable Chegg Com




Global Aromaticity In Macrocyclic Polyradicaloids Huckel S Rule Or Baird S Rule Accounts Of Chemical Research



How Does Huckel S 4n 2 Rule Determine Aromaticity Quora




130 What Is So Special About 4n 2 P Electrons Madoverchemistry
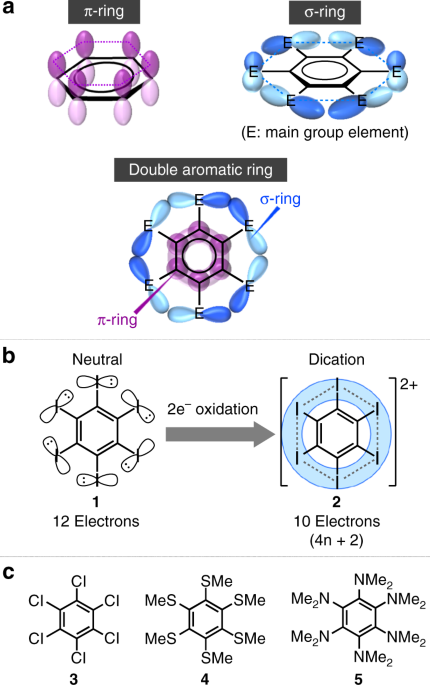



Double Aromaticity Arising From S And P Rings Communications Chemistry
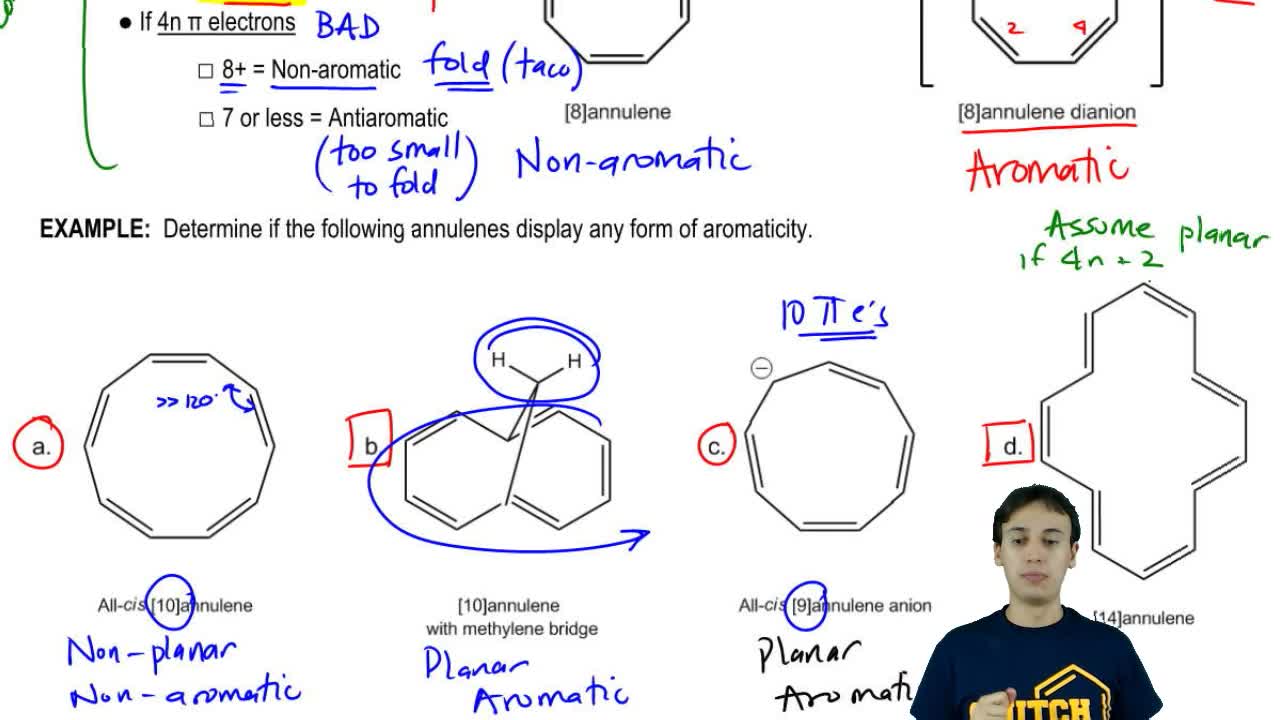



Annulene Organic Chemistry Video Clutch Prep




Aromaticity Mcqs For Neet With Answers
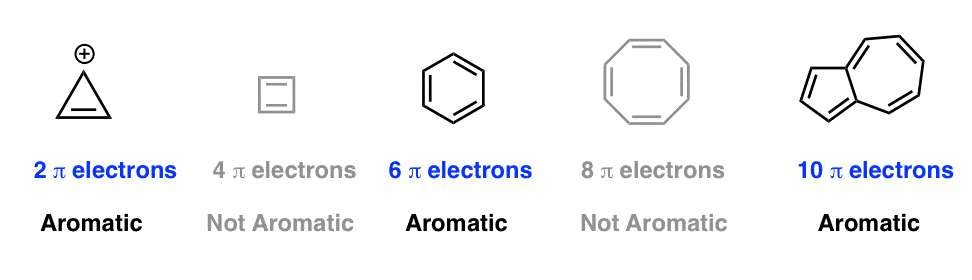



Huckel S Rule What Does 4n 2 Mean Master Organic Chemistry



What Are The Structures That Do Not Obey Huckel S Rule Of Aromaticity Quora




Aromatic Compound Characteristics Examples What Is An Aromatic Compound Video Lesson Transcript Study Com



2
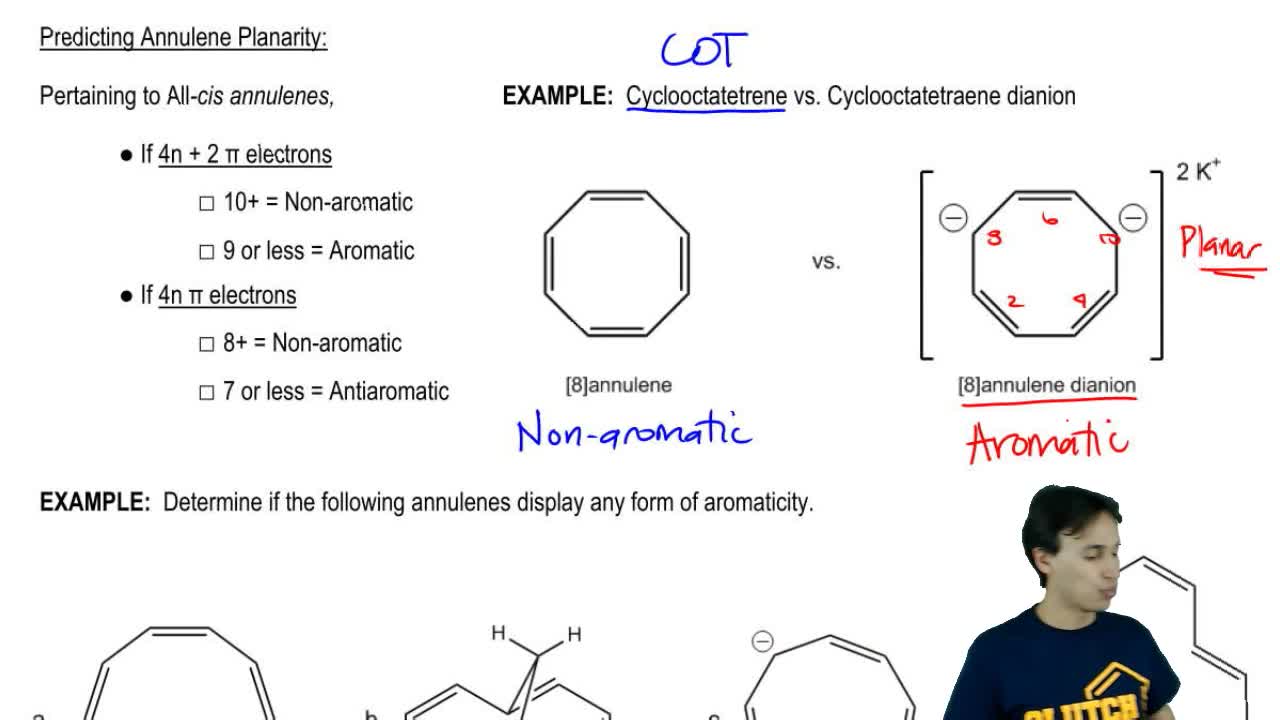



Annulene Organic Chemistry Video Clutch Prep




O Chem Part Ii Flashcards Quizlet




Unacademy India S Largest Learning Platform




Woodward Hoffmann Rules Wikipedia



Huckel S Rule Wikipedia




Aromatic Compound Characteristics Examples What Is An Aromatic Compound Video Lesson Transcript Study Com




Analysis Of Huckel S 4n 2 Rule Through Electronic Delocalization Measures The Journal Of Physical Chemistry A
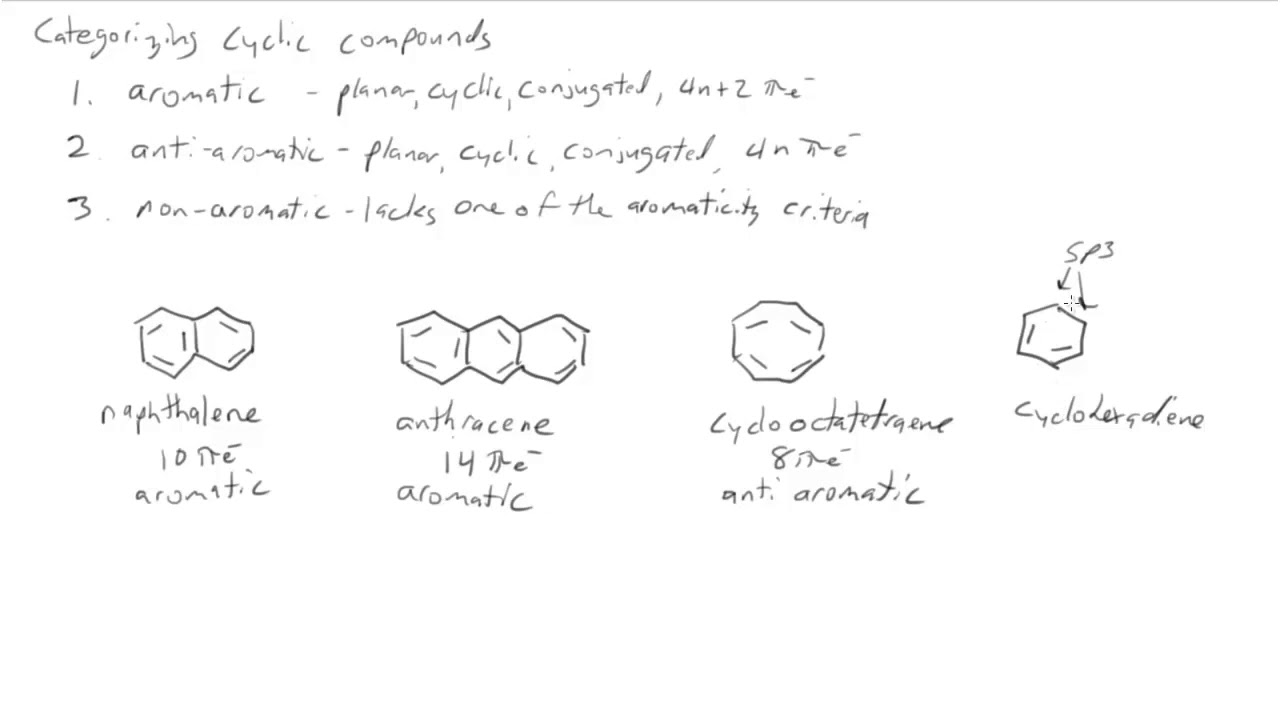



13 7 The Criteria For Aromaticity Huckel S Rule Chemistry Libretexts
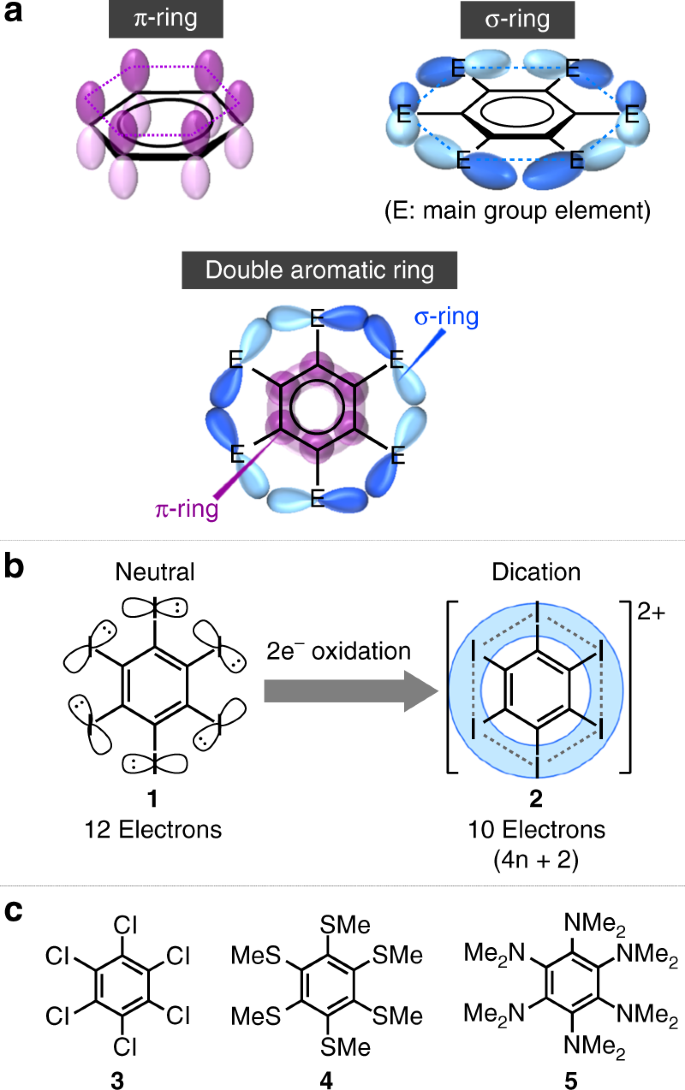



Double Aromaticity Arising From S And P Rings Communications Chemistry



Solved What Is The Value Of N From Huckel S Rule For The Following Aromatic Compound N 4 0 A 0 B N 2 0 6 N 9 0d N 8



Aromatic Stability Iii Video Khan Academy



2



15 3 Aromaticity And The Huckel 4n 2 Rule Chemistry Libretexts




Huckel S Rule For Aromaticity Time Saving Shortcut Youtube




A What Is The Value Of N In Huckel S Rule When A Compound H Quizlet



Rules For Aromaticity The 4 Key Factors Master Organic Chemistry




Why Is Cyclopropene Non Aromatic While It Has A 2 Pi Electron Quora
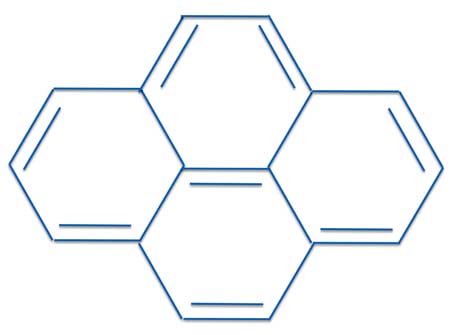



The Problem With Pyrene Michael J S Dewar To The Rescue




Chapter 17 Aromatic Compounds Aromatic Antiaromatic Or Nonaromatic Flashcards Quizlet



Aromatic Properties Mcat Question Of The Day
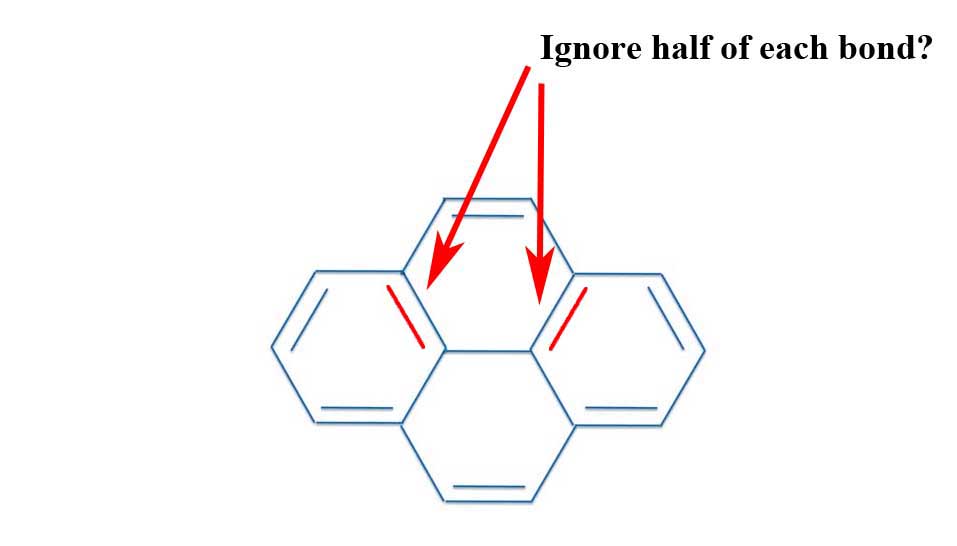



The Problem With Pyrene Michael J S Dewar To The Rescue



Q Tbn And9gct296etelqwagsbz2dhlt4k1b6 Ate25tcsgnhl74q454uc2rkpp7gz Usqp Cau




Ch 15 Benzene Reactivity Huckel S Rule 4n 2 P Electrons Aromatic Ppt Video Online Download
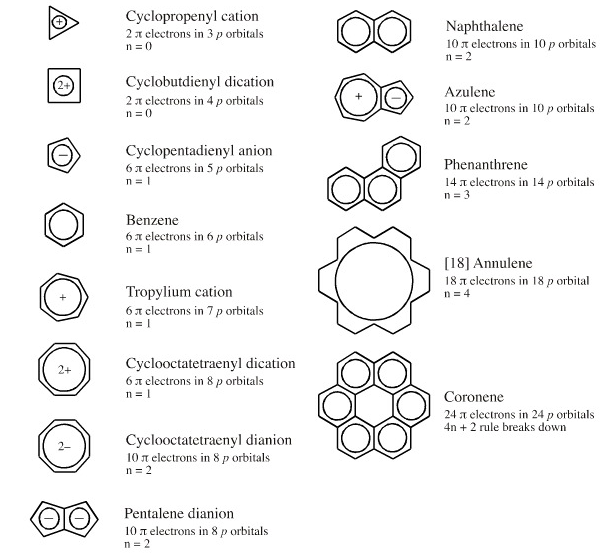



Lewis Theory Chemogenesis




Can Baird S And Clar S Rules Combined Explain Triplet State Energies Of Polycyclic Conjugated Hydrocarbons With Fused 4np And 4n 2 P Rings The Journal Of Organic Chemistry
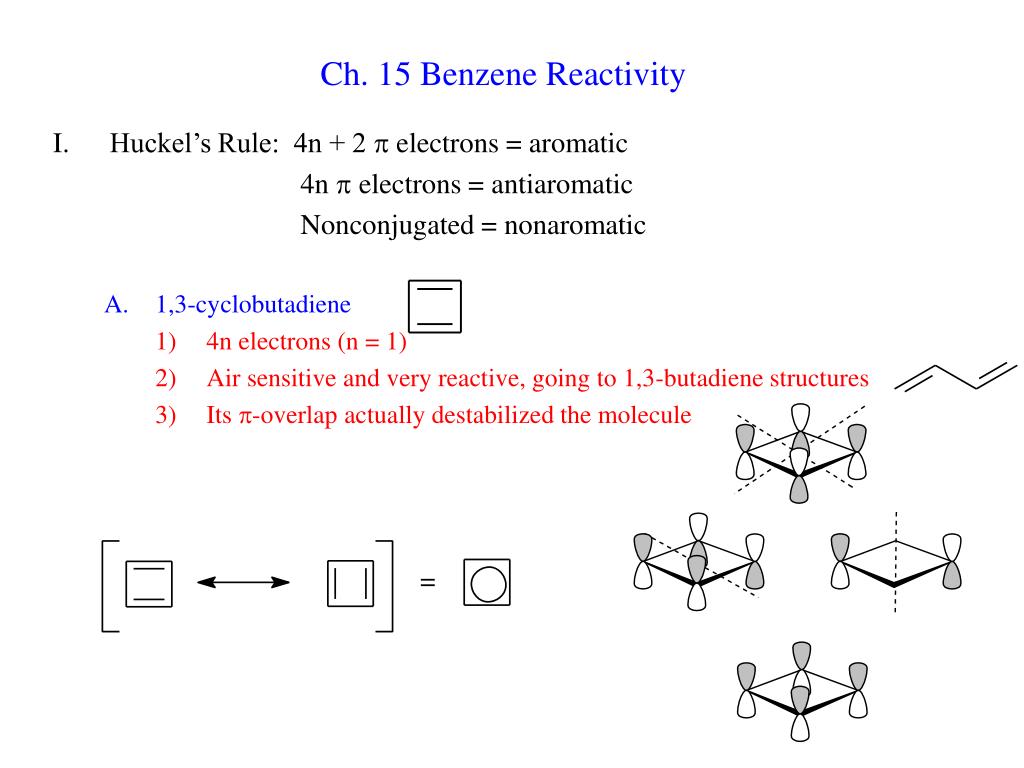



Ppt Ch 15 Benzene Reactivity Powerpoint Presentation Free Download Id




Aromatic Compound Characteristics Examples What Is An Aromatic Compound Video Lesson Transcript Study Com



Q Tbn And9gcqpb6zasp729jjrkmd2mpecmhmkzdrxx22fjiyxiddi5 Qpb Lxdujy Usqp Cau
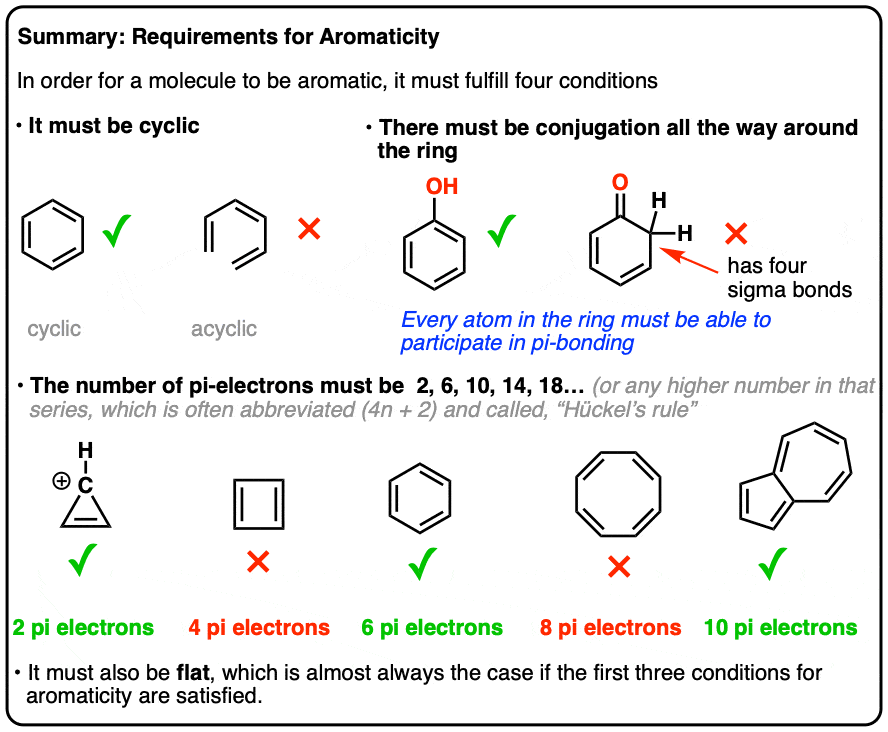



Rules For Aromaticity The 4 Key Factors Master Organic Chemistry



Illustrated Glossary Of Organic Chemistry Term
.jpg?revision=1&size=bestfit&width=317&height=292)



15 3 Aromaticity And The Huckel 4n 2 Rule Chemistry Libretexts



Despite Having A 4n Pi Electron System Why Is Cyclopentadiene Non Aromatic Quora




Aromatic Compound Characteristics Examples What Is An Aromatic Compound Video Lesson Transcript Study Com



How Does Huckel S 4n 2 Rule Determine Aromaticity Quora
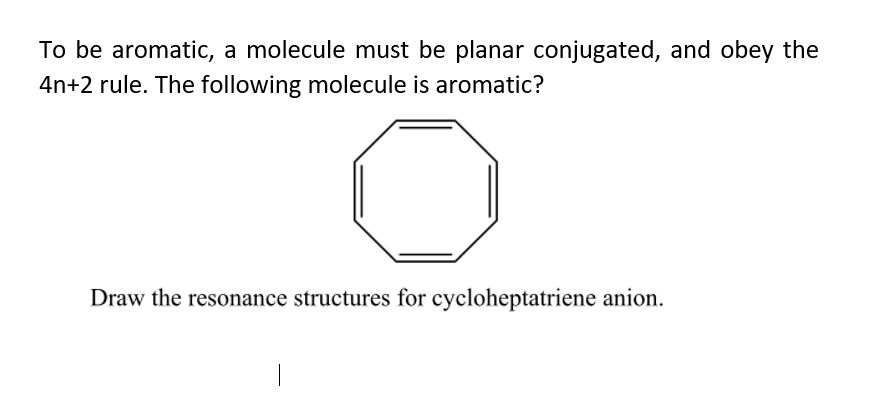



Answered To Be Aromatic A Molecule Must Be Bartleby




Aromatic Antiaromatic Or Nonaromatic Compounds Chemistry Steps



Aromaticity Meaning Classification Evidence Compounds Aesl
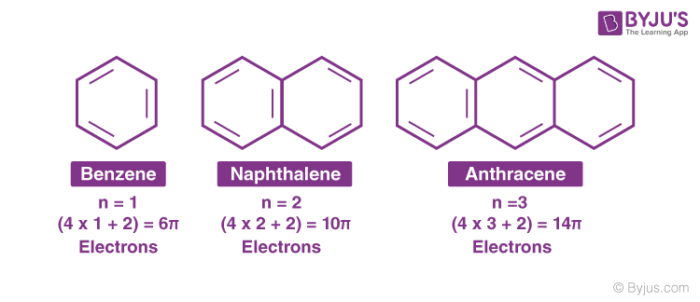



Aromatic Compounds Definition Example Properties Nomenclature With Videos




Notes On Huckel S Rule Unacademy




Aromatic Antiaromatic Or Nonaromatic Compounds Chemistry Steps



Illustrated Glossary Of Organic Chemistry Term




130 What Is So Special About 4n 2 P Electrons Madoverchemistry
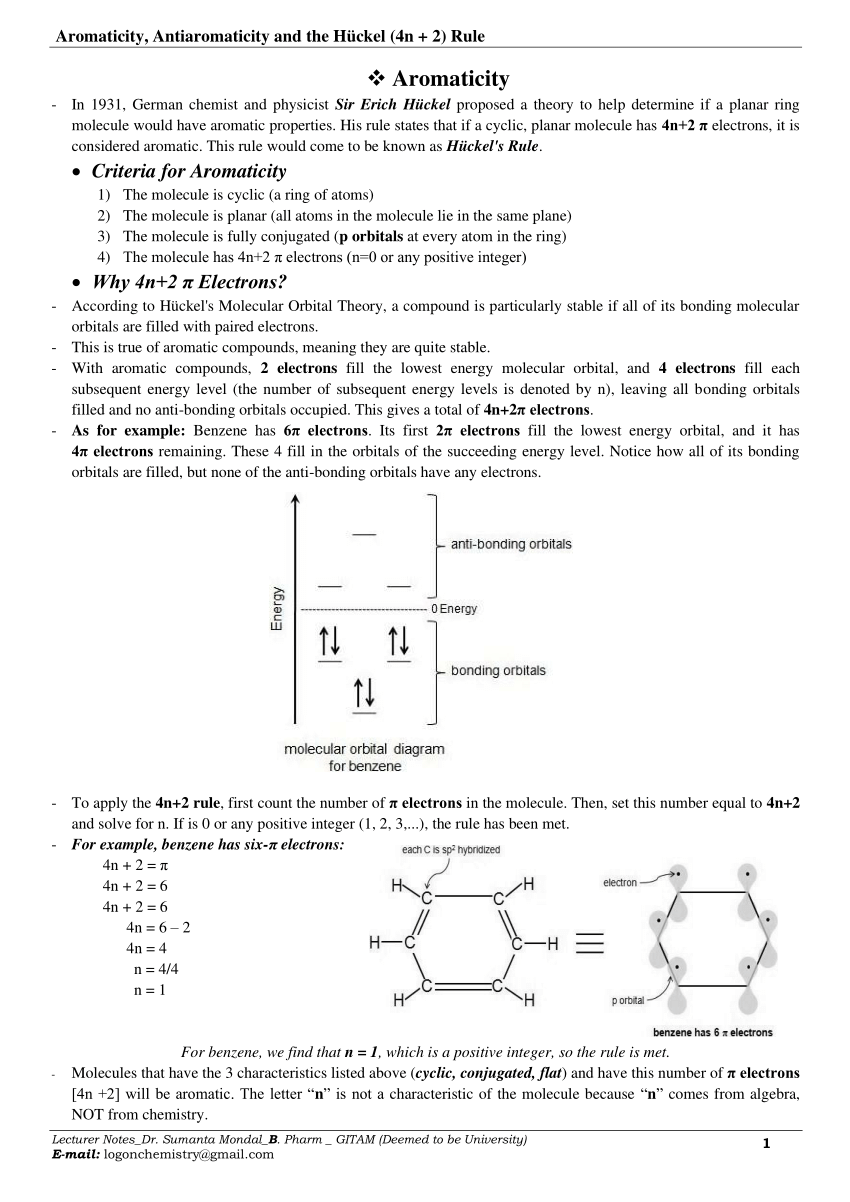



Pdf Aromaticity Antiaromaticity Homoaromaticity And The Huckel 4n 2 Rule
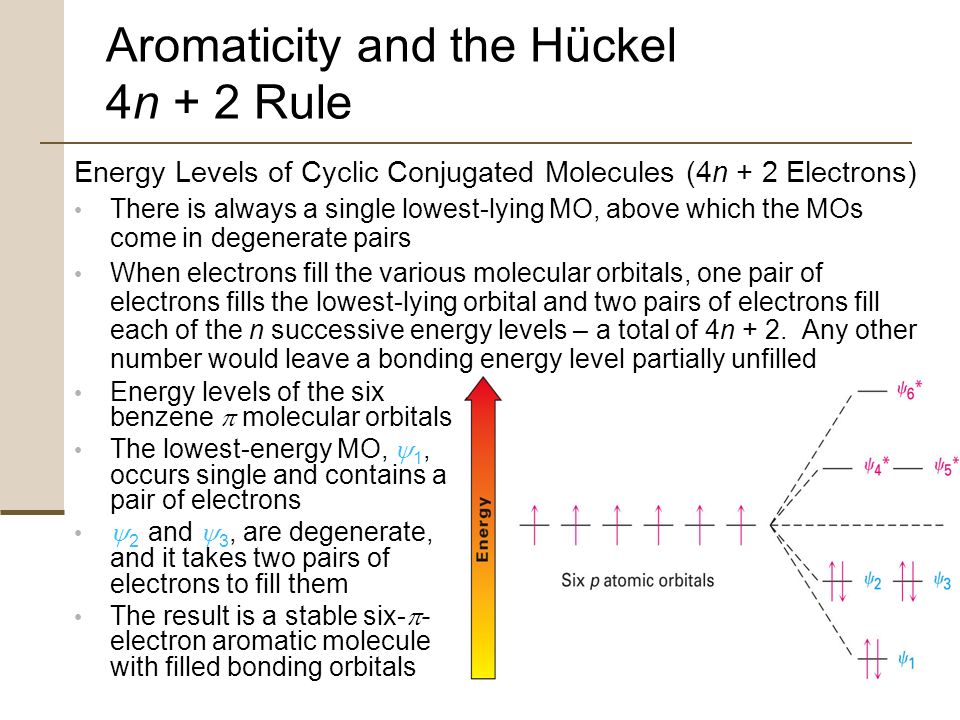



Chapter 8 Aromatic Compounds Ppt Download



2




Woodward Hoffmann Rules Wikipedia




Aromatic Hydrocarbons Organic Chemistry Video Clutch Prep
コメント
コメントを投稿